 Dementia - The Autophagic model
Dementia - The Autophagic model Dementia - The Autophagic model
Dementia - The Autophagic modelExamination of the processes that occur during the progressive destruction of the brain seen during the development of Alzheimer’s disease, could lead one to the generation of an Autophagic model of the disease. Autophagia (aw-t-uh-fe-jee-uh), is normally restricted to a process in which damaged organelles with the cell are slowly digested. There is, though, another type of autophagia in which bodily nutrition is maintained by sacrificial breakdown of some bodily tissues. Such a process could well be used to describe the series of events that occur in the brain, thereby resulting in the development of the characteristic morphology of the brain observed in AD.
In this model, in the prodromic phase of AD, the metabolism of glucose in the brain is affected, as would happen by a deficiency in vitamin B1, and/or vitamin B2. The result is that the favoured energy source of the brain, glucose is compromised, leading to lower energy production within the nerves of the brain and hence lower "processing speed".
1. Simple vitamin deficiency. Deficiency (ies) in vitamin B1 and/or vitamin B2 would result in reduced activity of the enzymes pyruvate dehydrogenase and also alpha-ketoglurate dehydrogenase (1,2,3). Altered glucose metabolism in AD is fairly well documented (4,5,6,7,8) , however, the cause of the altered metabolism is generally over-looked by most, with few citing B1 or B2 deficiency as being causal (1,2,3). Lack of activity of pyruvate dehydrogenase results in elevation of lactic acid (in straight B2 deficiency) and an elevation in pyruvate (in B1 deficiency), representing energy lost in glycolysis and increased excretion of these products which would later result in an increase of these easily assayable markers in urine.
2. Reduced Acetylcholine Production. A consequence of reduced activity of pyruvate dehydrogenase in the brain, is that pyruvate is not converted to acetyl-CoA, and so levels of acetyl CoA would be reduced, which would in turn reduce the production of acetyl-choline in the brain (also known to occur in AD). Lack of acetyl-choline in turn would lead to lack of production of acetyl-choline, and lack of acetyl-choline is linked to loss of cholinergic neurons, and subsequent memory and attention deficits, a feature of AD (9,10,11).
Apart from glycolysis, potentially, acetyl-CoA can be obtained from metabolism of fat, however this is not the preferred energy source in the brain and additionally, as in functional B2 deficiency you can’t burn fat for energy as each of the enzymes involved in fatty acid metabolism is dependent upon the active form of vitamin B2 (FAD), for its activity..
There is a third way to generate acetylcholine, and that is via the metabolism of glucogenic amino acids (but this is blocked due to lack of activity of pyruvate dehydrogenase), or via metabolism of the ketogenic amino acids, Leu, Lys, Phe, Trp, Tyr and Ile. Of these Leucine and Isoleucine (2 of the 9 essential amino acids) are branched chain amino acids. The metabolism of branched chain amino acids, however, requires the adenosylcobalamin-dependent enzyme, methylmalonyl-CoA mutase. In functional B2 deficiency the lack of activity of methylenetetrahydrofolate reductase (MTHFR) and methionine synthase reductase (MTRR) (both FAD/NAD(P)H dependent) means that one eventually (or the AD person specifically) becomes vitamin B12 deficient, and B12 deficiency is also known as a co-factor in dementia/AD (12-24). Potentially though it would be possible to still generate a small amount of acetyl-CoA from the other ketogenic amino acids, Lys/Phe/Trp and Tyr, and of these Phe, Trp and Tyr are all required to make neurotransmitters, dopamine, epinephrine, nor-epinephrine, serotonin and melatonin. Theoretically you could also still sacrifice the production of these neurotransmitter for energy production, but in methyl B12 deficiency and in iron deficiency (also common in AD), the activity of the critical Kreb cycle enzyme, aconitase is reduced as there is reduced synthesis of the iron-sulphur complex, and reduced aconitase activity has also been shown to be a feature of AD, with mini-mental score estimations paralleling the activity of aconitase (26).
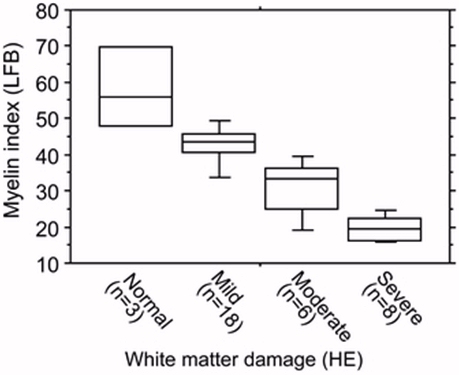
3. Breakdown of the Myelin Sheath. At this stage you have a brain desperate for energy and so it starts to “look” for new energy sources, and as it sits in a sea of fat, the lipids in the myelin sheath, and the accompanying myelin basic protein (MBP) it potentially could gain energy from either source. The brain “may know” that it can’t metabolize fat, but it should still be able to get energy from the MBP in the myelin sheath. So starts the loss of myelin in the brain, which also is typical of AD. This then would correlate with the autophagic stage of AD, hence the tag (27). Alterations in Tau, appear to post-date the decline in myelin sheath thickness.
In the autophagic stage, it is proposed that the brain tries to obtain energy from the break-down of the myelin sheath and the possible use of glycerol released from triglycerides, and the break-down of myelin basic protein to provide ketogenic and glucogenic amino acids for energy. Workers such as Klosinski and co-workers (28) have postulated that myelin is a potential ketogenic fuel supply for the brain, but in the absence of functional vitamin B2, the observed increase in short chain fatty acids, may more represent unmetabolized fats released due the break-down of the myelin sheath. As the autophagic state progresses there is a progressive loss of myelin thickness in the brain, which presumably accounts for the brain shrinkage seen in B12 deficiency and in the development of AD.
4. Weight Loss. Autophagia is not restricted to the break-down of myelin in the brain, and weight loss, frailty and cognitive decline are typical in the progression of dementia to AD (29-34). Not surprisingly, the rapidity of weight loss correlates with both cognitive decline and the reduced survival time (34, 35)). What is surprising is that individuals studying this phenomenon have failed to associate brain shrinkage, loss of myelin sheath and reduced energy output from mitochondria with paralleled frailty and weight loss.
5. Reduction in Vitamin B12 levels in the Brain. Accompanying the lack of vitamin B2 with the subsequent loss of functional vitamin B12, there is a reduction in the synthesis of melatonin (36) and adrenalin, which is inversely correlated with an increase in the neurotransmitter metabolites, HVA/VMA/QA and KA (37,38,39). Quinolinic acid (QA) is known to be neurotoxic and elevated QA have been associated with neurocognitive disorders, schizophrenia and depressive disorders (40). Elevated HVA has been implicated in neuronal toxicity and has been associated with motor impairment in Parkinson’s disease (41), and aggressive behaviour in frontotemporal dementia (38) and delirium (42).
|
|
|
| Deficiency of vitamin B12 (as judged by increasing MMA) is correlated with an increase in HVA. | Deficiency of vitamin B12 (as judged by increasing MMA) is correlated with an increase in QA. |
Apart from its known role in promotion of sleep, melatonin has also been implicated in neuronal maturation (43). Melatonin has also been shown to attenuate memory impairment in experimental models of AD (44), through stimulation of BDNF (Brain derived neurotrophic factor) and in inhibition of acetyl-cholinesterase production (45). Reduced daily melatonin production is associated with sleep disturbances and delirium in AD (46,47). Melatonin has also been shown to have a protective effect again production of the potentially toxic amyloid Aβ peptide (48) and against QA-mediated excitotoxic action. Lower production of adrenalin, due to lower levels of methyl B12, in turn can result in impaired thermoregulation seen in AD (49,50), as well as autonomic dysfunction and orthostatic hypotension, common conditions in AD and frontotemporal dementia (51), implicating the adrenergic system as being critically involved in the “sympathetic nervous system derangement” in the pathogenesis of AD (52).
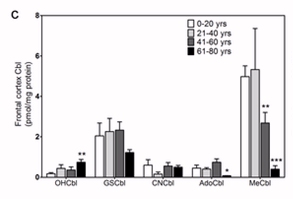
Change in vitamin B12 levels in the frontal cortex of the brain during aging. (Reproduced from Zhang etal, 2016(53)).
6. Reduction in the formation of Iron-sulphur proteins and decreased energy production. Essential in energy production are the iron-sulphur proteins, aconitase, succinate dehydrogenase and Complex II in the electron transport chain. Formation of the iron-sulphur clusters is dependent upon the movement of sulphur out of the methylation pathway into the sulphation pathway, and this only occurs when levels of S-Adenosylmethionine (SAM) are high enough to stimulate cystathionine beta synthase to process homocysteine. In low methyl B12, however, this does not happen and the activity of aconitase is greatly decreased in low iron and low vitamin B12. This leads to massive loss of energy from the body as the substrates citrate and succinate accumulate, ready for processing, but are eventually lost into urine. This loss of energy contributes to the ongoing fatigue in dementia and speeds up the rate of autophagia.
|
|
|
| Increasing MMA (a marker for reduced vitamin B12) is associated with increased loss of citrate in the urine. Representing a significant energy loss. | Reduced iron (carried on ferritin) contributes to the loss of activity of aconitase resulting in lower processing of citrate and incresed energy loss |
|
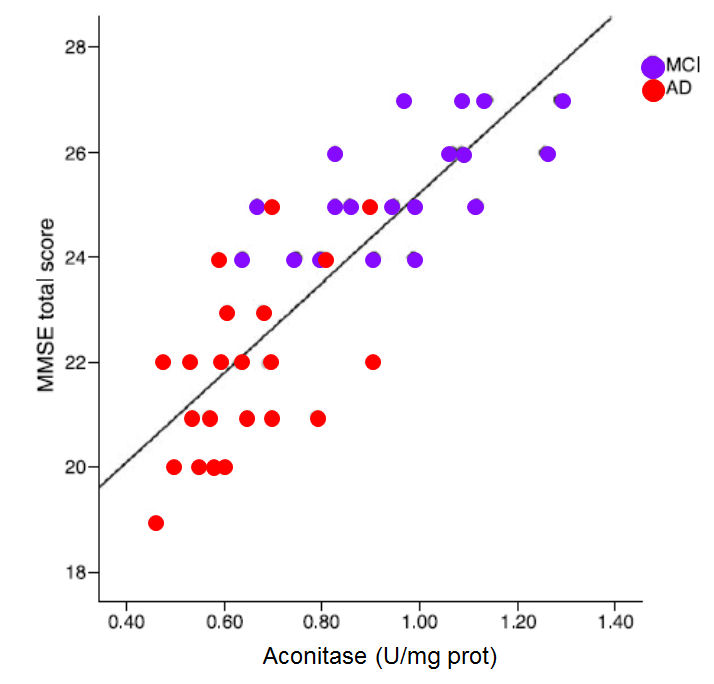
Energy production in the brain slowly decreases as the deficiency in methyl
B12 affects the transfer of dietary sulphur from methionine in the
methylation cycle through to the production of the iron sulfur proteins,
aconitase and succinate dehydrogenase, thus reducing their ability to
process citrate and succinate for energy. Reduced aconitase activity is a
feature of dementia, and this reduction correlates with decreased mini
mental score estimations in those with progressive dementia. (Figure. Data from Mangialasche etal, (26) |
 |
7. Reduction in the formation of the neurotransmitter, Hydrogen Sulfide. Apart from the reduced formation of iron-sulphur proteins observed in low vitamin B12 levels, there is an accompanying reduction in the formation of hydrogen sulfide in the brain. Hydrogen sulfide is a very important gaseous neurotransmitter normally produced in the brain, appears to have a role in repair in the brain and has has been shown to attenuate cognitive defects (54,55) and to reduce homocysteine induced pathology in AD (56), hence reduction in hydrogen sulfide production in the progressive B12 deficiency would effectively reduce its neuro-restorative capacity. It has also been implicated in brain synaptic remodelling (56,57) and inhibit amyloid formation (58), all of which would be reduced in the developing B12 deficiency in the brain.
8. Peripheral Neuropathy. The quest for food for the brain and nerves does not restrict itself to those of the central nervous system and loss of myelin in peripheral nerves leads to the characteristic symptoms of vitamin B12 deficiency being common viz: ataxia, loss of cutaneous sensation, muscle weakness, diminished or hyperactive reflexes, spasticity, urinary or faecal incontinence, orthostatic hypotension, loss of vision and hearing (59). Many of these symptoms though are often ascribed to old age, and the symptoms are over-looked as signs of the on-coming dementia..
9. Energy Loss. The prolonged deficiency in vitamin B2 and vitamin B12 leads to increasing loss of energy as fats are processed ready for consumption, yet the B2 deficiency prevents this and the prepared fatty acids are excreted in the urine. Similarly, lack of activity of pyruvate dehydrogenase leads to excretion of pyruvate and lactate into the urine. The inability to process glucogenic amino acids leads to further energy loss as proteins are catabolized and inability to process them for energy leads to increased excretion of glycerate and oxalate into the urine. Much is "brought to the table" but the majority of the prepared fatty acids, glycolysis products and amino acids are simply excreted into urine and contribute to elevated urinary organic acids, and resultant pH drop in urine and increased availability of these organic acids for digestion by urinary tract yeast and bacteria. Urinary tract infections are very common, more indications of altered metabolism.
10. Intestinal Absorption. With prolonged vitamin B12 deficiency, there is a failure in the maturation of the small intestinal mucosa, with the appearance of villous atrophy and failure of maturation of villous epithelial cells, which are normally responsible for specific uptake of nutrients and vitamins (60). Even if the desire was there, the body is slowly losing its capability to absorb the very vitamins that it needs for survival.
Finally the body exhausts itself of available energy for metabolism. The meal is over, there is no dessert, all the food has been eaten. The flame of life fizzles out, like the end of a sparkler. In a twist of fate, the body becomes its own “last supper”. Such was life, and such is death. RIP.

To some the above scenario may seem slightly over-dramatized, but in actuality the Biochemical parameters associated with the progression of Alzheimer's Disease fit the Autophagic Model very well.
The initiating factors in the progression of the disease may seem inconsequential at the time and may be a very small change in dietary intake, such as changing from iodized salt to more trendy Himalayan Rock salt (which despite its claims of mineral benefit has not iodine in it), changing from dairy milk (fortified with iodine in many countries) to soy milk (not fortified with iodine, and containing very little vitamin B2), changing from red meat to chicken (low in iron and vitamin B12), changing from gluten containing foods to gluten free (thus reducing the intake of vitamin B1, selenium and/or molybdenum), or even "going organic" (in many areas, such as the UK and many parts of Australia, the local soils are selenium deficient). Following these seemingly trivial events the story of decline begins. The disease may be very slow in onset and progress in an insidious fashion such that by the time symptoms become overt the initiating event is long forgotten.
The journey on the road to dementia can be a relatively long one. It is rather unusual in that no-body wants to go there, and the majority of people do not realize that they have started the journey. Most deny that they are "on the road" and what is more when they finally reach their destination, it is only the on-lookers and carers that realize that the destination has been reached. It is our hope that you do not start the journey, and if you do so, that you can recognize the signs and thereby get off the road and return to the start of the journey. Good luck to all, particularly the carers.
Gibson etal, 1998 Abnormalities of mitochondrial enzymes in Alzheimer disease PMID 9869323
Gibson etal, 2005 The alpha-ketoglutarate-dehydrogenase complex: ..... PMID 15953811
Gibson and Huang 2005 Oxidative stress in Alzheimer's disease.. PMID 15708429
Mosconi etal, 2008 Brain glucose hypometabolism and oxidative stress in preclinical Alzheimer's disease. PMID 19076441
Mosconi etal, 2009 FDG-PET changes in brain glucose metabolism from normal.... PMID 19142633
Moscone etal, 2010. Pre-clinical detection of Alzheimer's disease using FDG-PET PMID 20182125
Mosconi etal, 2013 Comparing brain amyloid deposition, glucose metabolism, and atrophy ..... PMID 23478305
Yao etal, 2011 Shift in brain metabolism in late onset Alzhemier's disease... PMID.22024209
Ferreira-Vieia etal, 2016 Alzheimer's disease: targeting the cholinergic system. PMID 26813123
Ricny 2013 Overlooked Alzheimer's smoking gun. PMID 23743622
Szutowicz etal, 2013 Acetyl-CoA the key factor for survival or death of cholinergic neurons... PMID 23677775
Wilson and Langman, 1966 Relation of sub-acute combined degeneration of the cord to vitamin B 12 deficiency. PMID 5988205
Oxnard and Smith, 1966 Neurological degeneration and reduced vitamin B12 in captive monkeys PMID 4960049
Green etal, 1975 Neurological changes in fruit bats deficient in vitamin B12 PMID 1118003
Zucker etal, 1981 B12 deficiency and psychiatric disorders: case report and literature review. PMID 703836
Weber etal, 1990 Nutritionally induced peripheral neuropathy PMID 2154308
Tangney etal, 2009 Biochemical indicators of vitamin B12 and folate insufficiency in cognitive decline PMID 19171834
Tangney etal, 2011 Vitamin B12, cognition and brain MRI measurements: a cross sectional examination. PMID 21947532
Skarupski etal, 2010 Longitudinal association of vitamin B-6, folate and B12 with depressive symptoms among older adults over time. PMID 20519507
Morris etal, 2005 Dietary folate and vitamin B12 intake and cognitive decline among community dwelling older persons. PMID 15824266
Morris etal, 2006 Thoughts on B-vitamins and dementia. PMID 16917152
Moore etal, 2016 A spectrum of exercise training.... PMID 26563933
McCaddon and Kelly 1994 Familial Alzheimer's disease and vitamin B12 deficiency. PMID 7976784
Wang etal, 2001 Vitamin B12 and folate in relation to the development of Alzheimer's disease PMID 11342684
Eussen 2006 Effect of oral vitamin B12 with or without folic acid on cognitive function.... PMID 16895884
Mangialasche etal, 2015 Lymphocytic mitochondrial aconitase activity is reduced in Alzheimer's disease and mild cognitive impairment. PMID: 25322927
Lim etal, 2016 Brain metabolism as a modulator of autophagy in neurodegeneration. PMID 26970520
Klosinski etal, 2015 White matter lipids as a ketogenic fuel supply in aging female brain PMID 26844268
LeBlanc etal, 2017 Weight trajectory over 20 years and likelihood of mild cognitive impairment.... PMID 27991654
Lee etal, 2017 Predictive factors against cognitive decline.... PMID 28436186
Spauwen etal, 2017 Associations of fat and muscle tissue with cognitive status PMID 28399220
Müller etal, 2017Association of exercise capacity and late onset dementia.... PMID 28082018
Franx etal, 2017 Weight loss in patients with dementia: Considering the potential impact of pharmacotherapy. PMID 28478593
Albala etal, 2017 Frequency of frailty and association with cognitive status...... PMID 28721027
Llibre etal, 2014 Frailty, dependency and mortality predictors in a cohort of Cuban older adults.... PMID 24487672
Sirin etal, 2015 Plasma 8-isoPGF2α and serum melatonin levels in patients with minimal cognitive impairment and Alzheimer disease. PMID 26838349
Vermeiren etal, 2013 Behavioral correlates of cerebrospinal fluid amino acid and biogenic amine neurotransmitter alterations in dementia.PMID 23159046;
Engelborghs etal, 2008 The dopaminergic neurotransmitter system is associated with aggression and agitation in frontotemporal dementia. PMID 18093695
Stuerenburg etal, 2004 5-Hydroxyindoleacetic acid and homovanillic acid concentrations in cerebrospinal fluid in patients with Alzheimer's disease, depression and mild cognitive impairment. PMID 15665806
Guillermin, GJ 2012 Quinolinic acid: neurotoxicity. PMID 22251552
Stefani etal, 2017Homovanillic acid in CSF of mild stage Parkinson's disease patients correlates with motor impairment. PMID 28108196
van der Cammen etal, 2006 Abnormal neurotransmitter metabolite levels in Alzheimer patients with a delirium. PMID 16955437
Chu etal, 2016 Effects of melatonin and its analogues on neural stem cells. PMID 26499359
Rudnitskaya etal, 2015 Melatonin Attenuates Memory Impairment, Amyloid-β Accumulation, and Neurodegeneration in a Rat Model of Sporadic Alzheimer's Disease. PMID 26402759
Shen etal, 2016 Effect of Melatonin and Resveratrol against Memory Impairment and Hippocampal Damage in a Rat Model of Vascular Dementia. PMID 28419991
Ángeles-Castellanos etal 2016 Loss of melatonin daily rhythmicity is asociated with delirium development in hospitalized older adults. PMID 28154742
Alagiakrishnan 2016 Melatonin based therapies for delirium and dementia. PMID 27355332
Mukda etal, 2016 Melatonin administration reverses the alteration of amyloid precursor protein-cleaving secretases expression in aged mouse hippocampus. PMID 27068758
Vandal etal, 2016 Impaired thermoregulation and beneficial effects of thermoneutrality in the 3×Tg-AD model of Alzheimer's disease. PMID 27555814
Hirakawa and Uemura, etal, 2012 Signs and Symptoms of Impending Death in End-of-life Elderly Dementia Sufferers: Point of View of Formal Caregivers in Rural Areas: -A Qualitative Study-. PMID 25649740
Struhal etal, 2016 Heart Rate Spectra Confirm the Presence of Autonomic Dysfunction in Dementia Patients. PMID 27567816
Femminella etal, 2016 Cognitive Function and Heart Failure: The Role of the Adrenergic System. PMID 27173155
Zhang etal, 2016 Decreased levels of vitamin B12 in aging, autism and schizophrenia. PMID 26799654
Chen etal, 2015 Neuroprotective effects of hydrogen sulfide and the underlying signaling pathways. PMID 25528761
Liu etal 2016 Hydrogen sulfide ameliorates learning memory impairment.... PMID 27883916
Kamat etal, 2015a Hydrogen sulfide ameliorates homocysteine-induced Alzheimer's disease-like pathology..... PMID 25019015
Kamat etal, 2015b Role of hydrogen sulfide in brain synaptic remodeling PMID 25747482
Rosario-Alomar etal, 2015. Hydrogen sulfide inhibits amyloid formation PMID 25545790
Healton etal, 1991 Neurologic aspects of cobalamin deficiency. PMID 1648656
Foroozan and Trier 1967 Mucosa of the small intestine in pernicious anemia. PMID 6038617
Copyright © 2016 preventingdementia.org. All Rights Reserved.
The statements on this site compose a compendium of generally recognized signs
of Alzheimer's disease. They also are formulated from a summary of relevant
scientific publications. In addition they may contain some forward looking
statements of a general nature.
Reproduction in whole or in part in any form or medium without express written
permission is prohibited